Impact of Cell Line and Reporter Gene Selection on In-Vitro mRNA-LNP Transfection Evaluation
- Apr 19
- 6 min read
Technical Review | AAPS Open, 2025 | Zhen, Lin, Darji, Dan, Ranjbar & Lu | Featured Tool: PreciGenome NanoGenerator Flex-M

Background: mRNA-LNP Transfection and Cell Line Selection
Messenger RNA lipid nanoparticle (mRNA-LNP) technology has emerged as a transformative platform for vaccines and therapeutics, validated by the clinical success of COVID-19 mRNA vaccines. With over 70 pipeline drug candidates currently in development across companies like Moderna, BioNTech, and Sanofi, mRNA-LNP therapies now target a wide range of indications beyond infectious disease — from protein replacement therapies and cancer immunotherapies to genome editing applications.
In-vitro transfection is one of the most critical methods for evaluating mRNA-LNP performance, as it measures the ability of LNPs to deliver mRNA into cells and produce functional protein. However, transfection efficiency is highly dependent on cell type and reporter gene selection. Primary mammalian cells, suspension cells, and adherent cells all respond differently to LNP-mediated delivery. Despite this, few studies have systematically examined how these choices affect the reliability and reproducibility of transfection assays.
In this study, Lu and colleagues at the University of Connecticut investigated how the choice of cell line (Jurkat, L-929, and HEK 293T) and reporter gene (firefly luciferase vs. eGFP) influences in-vitro transfection evaluation of mRNA-LNPs. Their findings provide practical guidance for developing robust and reproducible transfection assays — a critical need for the mRNA-LNP field.

Fig. 1. (A) Bioluminescence and (B) percent cell viability of Jurkat cells transfected with different concentrations of mRNA-LNPs across three independent batches. Jurkat cells displayed limited bioluminescence (8–270 RLU), non-linear dose response, and significant cytotoxicity at mRNA concentrations as low as 0.2 μg/mL. (Adapted from Zhen et al., AAPS Open, 2025)
Methodology
The researchers prepared mRNA-LNPs using the microfluidic mixing method with a PreciGenome NanoGenerator Flex-M (Precigenome LLC, San Jose, CA, USA). The lipid mixture consisted of SM-102, DSPC, cholesterol, and DMG-PEG 2000 at a molar ratio of 50:10:38.5:1.5 in anhydrous ethanol. The aqueous phase contained mRNA in 0.1 M citric buffer (pH 4.0). The two phases were mixed at a total flow rate (TFR) of 3 mL/min with a flow rate ratio (FRR) of 3:1 (aqueous:organic) at room temperature, yielding a final mRNA concentration of 25 μg/mL and total lipid concentration of 3.86 mg/mL before dialysis. The resulting LNPs were dialyzed against 20 mM Tris buffer (pH 7.4). Particle size distribution and polydispersity index were measured by dynamic light scattering (Zetasizer Pro), and encapsulation efficiency was quantified by the RiboGreen assay. Three cell lines were tested: Jurkat (suspension T lymphocytes), L-929 (adherent muscle fibroblasts), and HEK 293T (adherent epithelial cells). Transfection was assessed using two reporter mRNAs: firefly luciferase (quantified by bioluminescence assay) and eGFP (quantified by flow cytometry).
Jurkat Cells Show Limited Transfection and High Cytotoxicity
The Jurkat cell line, an immortalized human T lymphocyte line widely used in vaccine development studies, was the first model tested. Three independent batches of mRNA-LNPs were prepared with the NanoGenerator Flex-M, showing consistent PDI and encapsulation efficiency despite variations in particle size. Across all batches, Jurkat cells displayed limited bioluminescence (8–270 RLU) with no linear relationship between mRNA dose and luciferase expression. Bioluminescence peaked at approximately 2 μg/mL mRNA before plateauing or declining. More concerning, significant cytotoxicity was observed even at mRNA concentrations as low as 0.2 μg/mL, reducing cell viability to 75% and further to 40–50% at higher doses. The poor performance was attributed to the inherent difficulty of transfecting suspension cells, which interact with mRNA-LNPs in a more random and uncontrolled manner compared to adherent cell lines.
L-929 and HEK 293T Cells: Adherent Lines Outperform Suspension Cells
Given the challenges observed with Jurkat cells, the researchers evaluated two adherent cell lines. L-929 mouse muscle fibroblasts showed an increase in bioluminescence with increasing mRNA concentration up to approximately 3 μg/mL (maximum ~400 RLU), followed by a significant decline at higher concentrations — notably without any accompanying cytotoxicity. In contrast, HEK 293T cells exhibited a robust dose-dependent increase in bioluminescence across the entire tested range (0.02–4.60 μg/mL) without plateau or decline. Linear regression analysis yielded R² values of 0.9890, 0.9911, and 0.8828 for the three mRNA-LNP batches. HEK 293T cells also produced substantially higher bioluminescence levels than L-929 cells, with no cytotoxicity observed at any concentration tested.
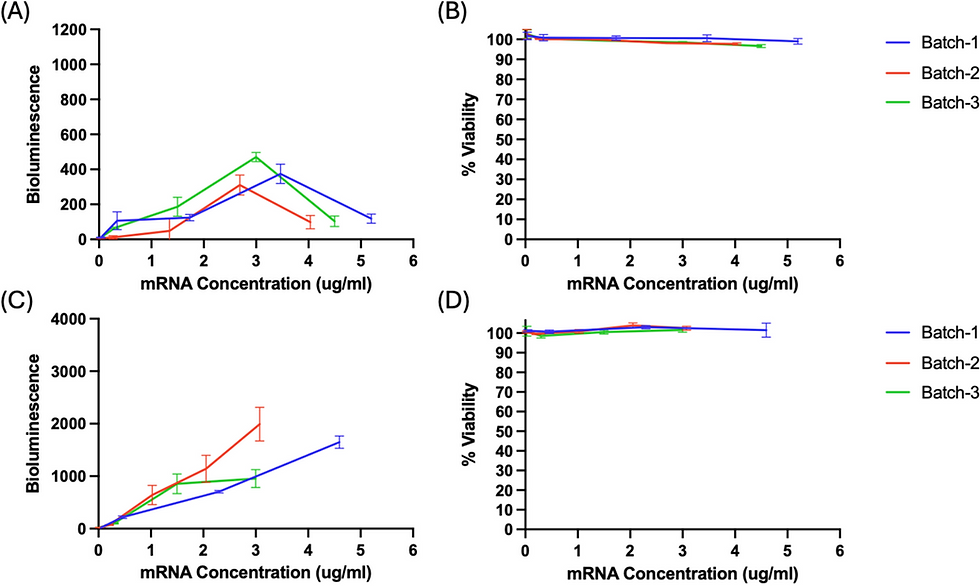
Fig. 2. Bioluminescence and percent cell viability of L-929 cells (A, B) and HEK 293T cells (C, D) transfected with different concentrations of mRNA-LNPs. L-929 cells showed limited signal that declined above 3 μg/mL. HEK 293T cells demonstrated a strong linear dose–response with high signal intensity and no cytotoxicity. (Adapted from Zhen et al., AAPS Open, 2025)
Luciferase Reporter Shows High Variability in HEK 293T Cells
Despite the favorable dose–response characteristics of HEK 293T cells with luciferase mRNA-LNPs, the researchers observed a critical limitation: high intra-group variability. When transfection was assessed in technical replicates of the same formulation, the coefficient of variation reached 58.59%. This high variability complicated interpretation of mRNA-LNP quality and raised concerns about the reproducibility of luciferase-based assays for formulation development. The bioluminescence signal fluctuated substantially among technical replicates, making it difficult to reliably distinguish between different formulations.
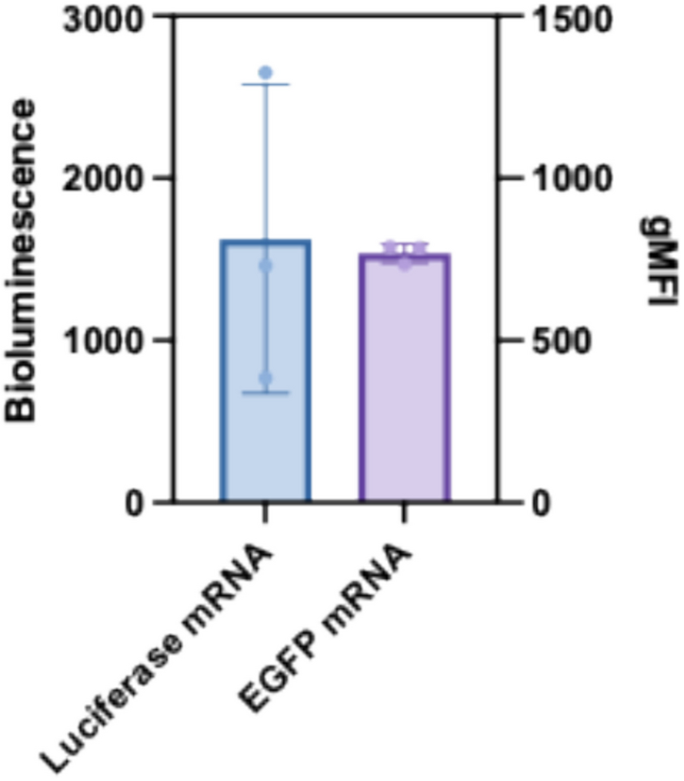
Fig. 3. Bioluminescence of HEK 293T cells transfected with luciferase mRNA-LNPs (left, blue) versus geometric mean fluorescence intensity (gMFI) of eGFP from HEK 293T cells transfected with eGFP mRNA-LNPs (right, purple). The luciferase assay showed substantially higher variability compared to the eGFP/flow cytometry method. (Adapted from Zhen et al., AAPS Open, 2025)
eGFP mRNA with Flow Cytometry Delivers High Reproducibility
To address the variability issue, the researchers switched to eGFP mRNA-LNPs quantified by flow cytometry. This approach enabled not only total protein expression analysis but also single-cell level analysis and distribution assessment within the cell population. Cell viability was not affected at 5 hours post-transfection and remained at 75–80% at 48 hours even at the highest concentrations. Critically, three key metrics — mRNA uptake (qRT-PCR), eGFP expression (gMFI), and percent eGFP-positive cells — all showed strong linear relationships with mRNA concentration (R² = 0.9786, 0.9706, and 0.9515, respectively). The coefficient of variation was less than 10.56% for mRNA uptake and less than 8.86% for eGFP-related measurements, a dramatic improvement over the 58.59% observed with the luciferase assay.
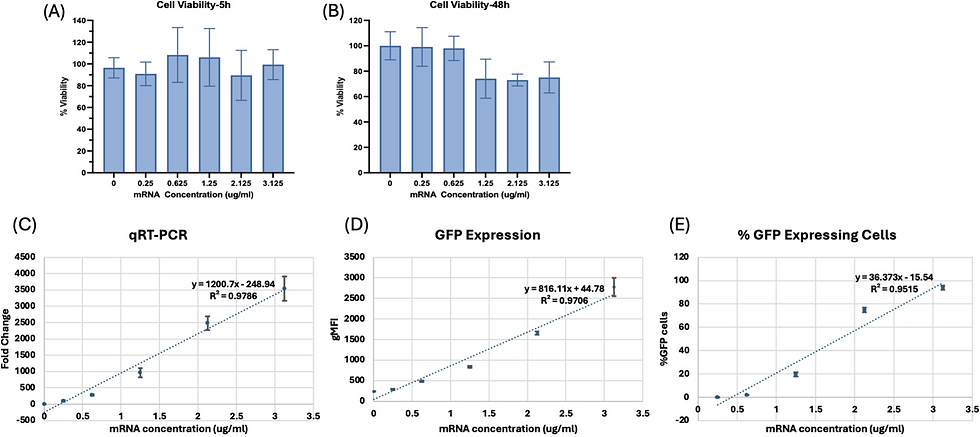
Fig. 4. Cell viability of HEK 293T cells at 5 hours (A) and 48 hours (B) after transfection, mRNA uptake by qRT-PCR (C), geometric mean fluorescence intensity of eGFP (D), and percentage of GFP-positive cells (E) with different concentrations of eGFP mRNA-LNPs. All three quantitative metrics showed strong linear dose–response relationships (R² > 0.95) with low variability (CV < 10%). (Adapted from Zhen et al., AAPS Open, 2025)
Key Findings at a Glance
Suspension cells are poor models for mRNA-LNP transfection — Jurkat cells showed limited protein expression (8–270 RLU), non-linear dose response, and significant cytotoxicity at low mRNA concentrations.
HEK 293T cells outperform L-929 cells as an adherent cell model — HEK 293T cells provided a strong linear dose–response (R² > 0.98), higher signal intensity, and no cytotoxicity across the tested concentration range.
Luciferase mRNA produces high assay variability — despite a good dose–response, the luciferase bioluminescence assay in HEK 293T cells showed a coefficient of variation of 58.59%, limiting its utility for reliable formulation screening.
eGFP mRNA with flow cytometry offers superior reproducibility — the eGFP/flow cytometry method reduced variability to less than 10% while maintaining strong linear correlations (R² > 0.95) across mRNA uptake, protein expression, and percent positive cells.
Single-cell resolution is a key advantage — flow cytometry-based eGFP quantification enables population-level, single-cell, and distribution analysis, providing richer data than bulk luminescence assays.
Cell line and reporter gene selection critically impact assay reliability — HEK 293T cells combined with eGFP mRNA-LNP represent a robust and reproducible model for in-vitro transfection evaluation.
PreciGenome NanoGenerator Flex-M — Featured Formulation Tool
All mRNA-LNPs in this study were prepared using the PreciGenome NanoGenerator Flex-M microfluidic mixing system. The NanoGenerator Flex-M enabled consistent and reproducible preparation of mRNA-LNPs with SM-102/DSPC/cholesterol/DMG-PEG 2000 (50:10:38.5:1.5 molar ratio) at a total flow rate of 3 mL/min and a flow rate ratio of 3:1. Despite preparing multiple batches at different scales, the system produced LNPs with consistent polydispersity and high encapsulation efficiency, demonstrating the reliability of microfluidic mixing for mRNA-LNP formulation development. The NanoGenerator Flex-M is a benchtop microfluidic platform designed for rapid, scalable LNP preparation — from screening to preclinical-scale production.
Reference
Zhen Z, Lin X, Darji M, Dan N, Ranjbar S, Lu X. "Impact of cell line and reporter gene selection on in-vitro transfection evaluation of mRNA lipid nanoparticles." AAPS Open. 2025;11:20. DOI: 10.1186/s41120-025-00125-3
Learn more about NanoGenerator Flex-M: www.precigenome.com


Comments