Organ-on-a-Chip (OOC) technology provides a novel in vitro platform with a possibility of reproducing physiological functions of in vivo tissue, more accurately than conventional cell-based model systems.
The technology opens up great opportunities for next-generation experiments of mimicking human organ functionality, microphysiology and morphology in vitro, replacing traditional animal-based model systems.
By realizing different organ functions on a chip, Organ-on-a-Chip technology is potentially useful for building models of complex diseases. The technology can also be used to study pharmacokentic model when new drugs are being developed.
PreciGenome offers customizable Organ-on-Chips system, which is able to recreate the dynamic in-vivo conditions for modeling biochemical and biophysical features of cells' native environment. Combining the PG-MFC flow controller and microfluidic membrane chips, the system provides multi-channel perfusion or reagent recirculation capability to ensure cell nurturing over weeks.
Organ-on-a-Chip System with Flow Controller
System Overview:
PreciGenome Organ-on-a-Chip system with PG-MFC controller provides both vacuum and pressure and makes it perfectly suited to organ-on-a-chip applications.
Our system provides culture media recirculation and multi-channel capability to ensure cell nurturing over days.

System Benefits:
-
Controlled media flow rates and shear stress
-
Long duration experiments
-
Automation
-
3D cell culture
-
Ready to connect with the incubator
-
Temperature control module is also available to integrate into the system
-
OEM and custom design available
Organ Models
The iFlow OOC System is validated across the full spectrum of microphysiological tissue models. Select any model to explore supported applications and chip configurations.

Cancer-on-Chip
Recapitulate the tumor microenvironment with perfusable vascularized tumor constructs for drug screening, immunotherapy evaluation, and metastasis modeling.

Blood-Brain Barrier on-Chip
Establish a functional BBB with tight junction integrity, astrocyte support, and selective permeability assays for CNS drug delivery and neuroinflammation research.

Spheroid-on-Chip
Perfuse 3D tumor spheroids and patient-derived organoids with controlled nutrient, oxygen, and drug gradients for high-content efficacy and toxicity profiling.

Gut-on-Chip
Model intestinal absorption, permeability, and microbiome interactions with physiological peristaltic-like flow and villi morphology under continuous perfusion.

Intestine-on-Chip
Model the large intestinal epithelium with crypt-villus architecture, mucus secretion, and commensal microbiota under anaerobic-aerobic interface conditions.

Bone Marrow-on-Chip
Model hematopoiesis, stem cell niche interactions, and myelotoxicity within a perfused 3D marrow matrix for radiation injury and chemotherapy toxicity studies.

Liver-on-Chip
Maintain long-term hepatocyte function and zonation under physiological oxygen and nutrient gradients for drug metabolism, hepatotoxicity, and DILI studies.

Kidney-on-Chip
Recapitulate proximal tubule reabsorption, glomerular filtration, and nephrotoxicity under physiological shear stress for renal drug toxicity and disease modeling.

Lymphoid-on-Chip
Recreate secondary lymphoid tissue architecture with T/B cell compartments, germinal center reactions, and antigen presentation for immunotherapy and vaccine research.

Lung-on-Chip
Recreate the alveolar air-liquid interface with mucociliary differentiation and pulmonary vascular perfusion for respiratory disease and toxicology studies.

Vasculature-on-Chip
Form perfusable 3D microvascular networks under physiological wall shear stress for thrombosis, angiogenesis, atherosclerosis, and vascular inflammation studies.

Other Organ-on-Chip Models
Don't see your organ model? The iFlow platform's chip-agnostic architecture supports any custom chip design. Our applications team configures validated workflows for your specific tissue.
Microfluidic Chips for Organ-on-a-Chip
The chips contain two cavities, each comprising an integrated membrane serving as cell culture area. Cells cultured on the membrane are perfusable independently from both the apical or basal side through upward directed (upper perfusion) and downward directed (lower perfusion) channels. The chips have two in- and outlet ports above and below the membrane.
The chip is made by injection molding from polystyrene (PS) or Topas. A 12 μm thick polyethylene terephthalate (PET) membrane (TRAKETCH) with a pore diameter of 8 μm or 0.2 um and a pore density of 1X10e5 pores/cm² was integrated in the upper and lower part of the chip by heat-sealing with the bulk material. This allows for organ-on-a-chip experiments such as small molecule transfer measurements, on-chip dialysis or cell culture experiments.




Image of the OOC membrane chip
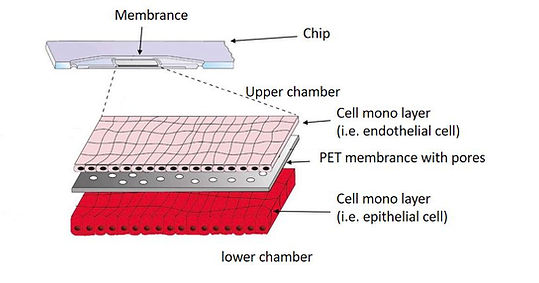
Image of porous PET membrane layer of the chip
Chips and channel structures were sealed on the top and bottom sides with a bonding foil using a low temperature bonding method.
Oxygen plasma treatment for hydrophilization of the whole chip surface can be performed to support cell adhesion and to reduce air bubble formation in the chips (Reference [1]).
Culture Modes and Applications
Mode #1: Single-sided Culture
A cell monolayer is cultivated on one side of the membrane in the chip.

Application Examples of Single-sided Culture
-
Skin-on-a-Chip Model
Skin Models with air-liquid interface are a good fit for this culture mode. Skin-on-a-chip (SoC) are used for testing of topical pharmaceuticals and cosmetics, studying the pathology of skin diseases and inflammation. Also it is to test for the presence of antigens or antibodies which could denote the presence of a pathogen.
The model in the figure shows a polarized skin cell monolayer is cultured on one side of the membrane with cells exposed to air in the upper chamber. The flow of culture medium goes through the lower chamber.

-
Apical-Basal Cell Polarity Assay Model
A cell monolayer is cultivated at one side of the membrane. On the other side, a 3D gel matrix fills the chamber. In some applications cells are embedded in the 3D gel matrix. Chemical factors inside the 3D gel matrix lead to the polarization of a cell monolayer at the opposite side of the 3D gel matrix.

Mode #2: Double-sided Culture
Two cell monolayers, one layer on the top of membrane and another layer beneath the membrane, are cultivated simultaneously in the chip. Molecules are transferable through the pores on the membrane. Thus, this culture mode is fit for cell studies such as signaling, co-culture, and transport studies.

Application Examples of Double-sided Culture
-
Lung-on-a-Chip Model
The lung-on-a-chip is a complex, three-dimensional model of a living, breathing human lung on a microchip. The device is made using human lung and blood vessel cells and it can predict absorption of airborne nanoparticles and mimic the inflammatory response triggered by microbial pathogens. Lung-on-a-chips are being designed in an effort to improve the physiological relevance of existing in vitro alveolar-capillary interface models. Such a multifunctional microdevice can reproduce key structural, functional and mechanical properties of the human alveolar-capillary interface.
In this example (reference [2]), a lung-on-a-chip model establishes a human in vitro alveolus model system, consisting of vascular and epithelial cell structures with cocultured macrophages. The infection of epithelial cells induced a high inflammatory response that spread to the endothelium. The system demonstrated significant endothelial cell damage associated with loss of barrier function. It can be used as an immune-responsive model reflecting the complex crosstalk between pathogens and host. This is a powerful system for mechanistic studies of host-pathogen interactions and the identification of molecular and cellular targets of novel treatment strategies in pneumonia.

Example of Lung on a Chip model with alveolar epithelial cells in air phase in the upper chamber and endothelial cells cocultured with perfusion medium in the lower chamber. Figure is from reference [2].
System Setup Example
- Lung-on-a-Chip
In this lung-on-a-chip case, two pressure and one vacuum sources are used which connect to different inlets of a chip with the controller. PG-MFC controller has multiple sources of vacuum and pressure. It meets most requirements of organ-on-a-chip applications.
Two pressure lines push different culture media to deliver into the chip to mimic blood flow into lung and exchange chemicals through cell channel bilayer. The vacuum line connects to the side chamber to simulate the breathing process in a lung.

Example of sine output on vacuum Line, which is used to actuate the membrane between the top and bottom chambers.
Meanwhile, the airflow rate is also monitored in realtime to indicate the leakage in the flow.
-
Other intended applications
-
Brain-on-a-chip
-
Heart-on-a-chip
-
Kidney-on-a-chip
-
Nephron-on-a-chip
-
Vessel-on-a-chip
-
Intestine-on-a-chip
References
-
Raasch, M.; Rennert, K.; Jahn, T.; Peters, S.; Henkel, T.; Huber, O.; Schulz, I.; Becker, H.; Lorkowski, S.; Funke, H. Microfluidically supported biochip design for culture of endothelial cell layers with improved perfusion conditions. Biofabrication 2015
-
Deinhardt-Emmer S, Rennert K, Schicke E, Cseresnyes Z, Windolph M, Nietzsche S, Heller R, Swiczak F, Haupt KF, Carlstedt S, Schacke M, Figge MT, Ehrhardt C, Löffler B, Mosig AS; Co-infection with Staphylococcus aureus after primary influenza virus infection leads to damage of the endothelium in a human alveolus-on-a-chip model., Biofabrication, 2020













